AREDS3 and multivitamin coadministration: practical safety, rationale, and clinical guidance for eye care professionals
By Julie Poteet, OD; Jessilin Quint, OD; and Shane R. Kannarr, OD
The contents of this article are informational only and are not intended to be a substitute for professional medical advice, diagnosis, or treatment recommendations. This editorial presents the views and experiences of the author and does not reflect the opinions or recommendations of the publisher of Optometry 360.
A new supplement (PreserVision® AREDS3; Bausch + Lomb, Bridgewater, NJ) that combines the nutrients in AREDS2 (PreserVision AREDS2) with a B-vitamin complex has recently become available, and a clinical trial is planned.1 The addition of B vitamins to the new formula is intended to support cellular metabolism and maintain healthy levels of homocysteine, a non-proteinogenic amino acid that can harm ocular health when present at high levels.2-6 Accordingly, the new formula may benefit a wider range of patients than AREDS2, such as those with early-stage age-related macular degeneration (AMD).1
Given the common usage of multivitamins among US adults,7,8 practical guidance is warranted regarding the coadministration of AREDS3 with multivitamins. Here, we describe the rationale for the inclusion of B vitamins in AREDS3, discuss safety considerations for use of AREDS3 in combination with a multivitamin, and provide clinical guidance for coadministration.
AREDS3: Advancing Beyond AREDS2
AREDS3 represents an evolution informed by mechanistic science and nutritional epidemiology. A growing literature highlights the importance of homocysteine homeostasis on ocular health. Excessive accumulation of homocysteine can trigger inflammation-related oxidative stress.2 Elevated homocysteine has been implicated in apoptosis of lens epithelial cells,3 retinal ischemia and neovascularization,2 disruption of retinal pigment epithelial (RPE) structure and function,4 and damage to vascular endothelial cells and the optic nerve.2,5,6
Several B vitamins serve as cofactors in homocysteine metabolism. Vitamins B9 and B12 help convert homocysteine into methionine via the enzyme methylenetetrahydrofolate reductase (MTHFR),2,9,10 while B6 facilitates its conversion into cysteine. When levels of these vitamins are insufficient, or when MTHFR gene polymorphisms impair metabolism, homocysteine accumulates,2,9 with downstream consequences for ocular health. Conversely, boosting intake of vitamins B6, B9, and B12 has been shown to reduce homocysteine concentrations.11,12
Lowering homocysteine levels is a key mechanism via which B vitamins are believed to reduce vascular damage and protect the integrity of the RPE.1 Additional protective mechanisms of B vitamins in AMD include maintaining energy metabolism and normal mitochondrial function13,14; mitigating oxidative stress via antioxidant effects10,15; exerting neurotrophic effects important for maintaining nerve function16,17; improving RPE integrity and health by inhibiting epithelial-to-mesenchymal transition18; and suppressing production of AMD/drusen-associated proteins, inflammatory and complement factors, and vascular endothelial growth factor.19,20
National Health and Nutrition Examination Survey data confirm that B vitamin insufficiency remains common in older adults. Approximately 10% to 20% of those not using multivitamins have insufficient intake of key B vitamins.21,22 While supplements substantially reduce the prevalence of B vitamin inadequacies,21 older adults with low income, from minority backgrounds, or with obesity may be at increased risk.23,24 Even older adults with normal serum B vitamin levels may have elevated homocysteine, suggesting that serum vitamin concentrations can underestimate tissue-level deficiency.25
Eye care providers (ECPs) sometimes ask whether supplementation at supratherapeutic levels is restoring a deficiency or delivering a “super dose.” The doses of B vitamins in AREDS3 reflect levels shown to be therapeutically meaningful in the context of AMD.26 Doses of vitamins B6, B9, and B12 in AREDS3 are grounded in the Women’s Antioxidant and Folic Acid Cardiovascular Study (WAFACS), a large, randomized, double-masked, placebo-controlled trial in which a B6/B9/B12 supplement reduced AMD risk by 34% over an average duration of 7.3 years.27 In WAFACS, approximately 23% of participants in each treatment group were taking a multivitamin at baseline. However, participants were not permitted to take vitamin B supplements or multivitamins exceeding the recommended daily allowance (RDA) of vitamins B6, B9, or B12. Thus, background intake of B vitamins was controlled and relatively modest.
Exact B Vitamin Levels and Percent RDA
ECPs appropriately want precise dosing data presented in a meaningful safety context. AREDS3 provides vitamin B6 at 50 mg (2941% of the RDA of 1.7 mg), vitamin B9 as 0.9 mg of folic acid (225% of the RDA of 400 μg), and vitamin B12 at 1 mg (41,667% of the RDA of 2.4 μg), alongside B1, B2, B3, B5, and B7 at doses also exceeding their respective RDAs (see Table 1). These percentages appear large, but the RDA is the intake sufficient to meet the nutritional requirements of nearly all healthy individuals and is designed to prevent deficiency rather than define safety.28
Table 1. Comparison of Formulation-Specific Vitamin B Quantities in AREDS3 and Centrum Silver Women 50+ With Recommended Dietary Allowance, Tolerable Upper Intake Levels, and Toxicity Thresholds
| B Vitamin (Form) | AREDS3 Formulation-Specific Quantity (2 soft gels, mg) (% RDA) | Centrum Silver Women 50+ Formulation-Specific Quantity (1 tablet, mg) (% RDA) | RDA (mg)‡28 | UL (mg)28 | Toxicity Threshold (mg)10,31 |
| B1 (thiamine mononitrate) | 3.6 (300) | 1.1 (92) | 1.2 | NA | NA |
| B2 (riboflavin) | 3.9 (300) | 1.1 (85) | 1.3 | NA | NA |
| B3 (niacin/niacinamide) | 32* (200) | 14† (88) | 16 | 35§ | 3000 |
| B5 (calcium pantothenate) | 15 (300) | 5 (100) | 5 | NA | NA |
| B6 (pyridoxine hydrochloride) | 50 (2941) | 5 (294) | 1.7 | 100 | 1000 |
| B7 (biotin) | 0.09 (300) | 0.03 (100) | 0.03 | NA | NA |
| B9 (folic acid) | 0.9 (225) | 0.4 (100) | 0.4 | 1.0 | N/A |
| B12 (cyanocobalamin) | 1.0 (41,667) | 0.05 (2083) | 0.0024 | NA | NA |
| *Niacinamide.
†Niacin. ‡RDAs shown are for men ≥51 years of age. Percent RDAs for AREDS3 and Centrum Silver Women 50+ are based on these RDA values. §The UL of 35 mg is based on niacin. Even though the combined intake of vitamin B3 (niacinamide) in AREDS3 and a multivitamin exceeds 35 mg, it is well within the known safety limit for niacinamide (<1500 mg). Vitamin B9 (folic acid) is considered nontoxic, although high-dose folic acid supplementation (>1 mg/day) can mask the neurological signs of vitamin B12 deficiency30 NA, not applicable; RDA, recommended dietary allowance; UL, tolerable upper intake level. |
|||||
RDA Versus Tolerable Upper Intake Level and Toxicity Threshold
A common source of concern among ECPs is the conflation of RDA exceedance with risk. In practice, the upper intake level (UL) is the relevant safety reference. The UL provides a safety ceiling as the highest level of intake that is likely to present no risk of adverse health effects based on review of adverse events data.28 Thus, even daily intake at the UL is unlikely to cause adverse health effects in almost all individuals in the general population. The UL includes a safety margin and is usually well below true toxicity levels. Exceeding the UL does not mean toxicity will occur, only that the risk begins to increase. Additionally, the safety margin between the RDA and the UL is typically wide. Standard over-the-counter multivitamins routinely deliver numerous nutrients above their RDAs,29 and therapeutic formulations designed for clinical effect do the same by design. Nutrient toxicity generally requires chronic high exposure.28
Because B-complex vitamins are water-soluble, excess amounts are readily excreted renally rather than accumulating in tissue stores, further broadening the practical safety margin.10 Vitamins B1, B2, B5, B7, and B12 have no established UL (see Table 1) and are considered essentially nontoxic even at high supplemental doses. The UL for vitamin B3 is 35 mg, which is primarily based on adverse effects (eg, flushing, hepatotoxicity) associated with high-dose niacin (>3 g/day [toxicity threshold]). In contrast, use of the niacinamide (also called nicotinamide) form of vitamin B3 in doses of up to 1500 mg/day is considered safe for healthy individuals.30 Chronic vitamin B6 supplementation exceeding 1 g/day (toxicity threshold) has been associated with sensory neuropathy and ataxia, and its UL for adults is 100 mg/day.10,31 Vitamin B9 is considered nontoxic, although high-dose folic acid supplementation (>1 mg/day) can mask the neurological signs of vitamin B12 deficiency, allowing neurological damage to progress undetected.10 Additionally, high intake of synthetic folic acid may result in circulating unmetabolized folic acid, although the clinical significance of this finding remains an area of ongoing investigation.
Coadministration With Multivitamins: Safety and Interaction Considerations
Coadministration of an AREDS-based supplement with a multivitamin is not a new scenario. In the landmark AREDS2 trial, almost 90% of participants were already taking multivitamins at baseline.32 Participants in the AREDS clinical program were offered Centrum Silver to standardize non-study supplement use. No safety signals emerged from combined use. Furthermore, AREDS2 launched and gained widespread clinical adoption ahead of the publication of extended outcome data in 2022,33 establishing a precedent for thoughtful, evidence-informed advancement of supplement-based prevention strategies.
When AREDS3 is combined with a standard multivitamin, such as Centrum Silver Women 50+, most cumulative B vitamin intakes remain well within established safety thresholds (see Figure 1). The following summarizes key nutrient-level considerations:
The form of vitamin B3 in AREDS3 is niacinamide (nicotinamide), while the UL of 35 mg is based on niacin. Even though the combined intake of vitamin B3 in AREDS3 and a multivitamin exceeds 35 mg (see Figure 1), it is well within the known safety limit for niacinamide (<1500 mg).30 The AREDS3 dose of vitamin B6 (50 mg) represents only 50% of the UL (100 mg; see Table 1). The typical multivitamin adds only a modest dose of vitamin B6 (see Figure 1), such that combined intake with AREDS3 would remain comfortably below the UL. The 1 mg/day UL for folate applies specifically to synthetic folic acid, given concerns that unmetabolized folic acid can mask the hematological signs of vitamin B12 deficiency.10,28 When AREDS3 is combined with a multivitamin, the daily intake of folic acid may exceed the UL (see Table 1 and Figure 1). Since AREDS3 provides high-dose vitamin B12 supplementation (see Table 1), vitamin B12 deficiency is unlikely to be a problem except in rare cases when the individual has a functional B12 deficiency. No established UL exists for B12 (see Table 1). It has a well-documented low toxicity profile, and any excess is readily excreted.10
Figure 1. AREDS3 vitamin dosing in relation to a typical multivitamin (Centrum Silver Women 50+) and tolerable upper intake levels for vitamins B3, B6, and B1228
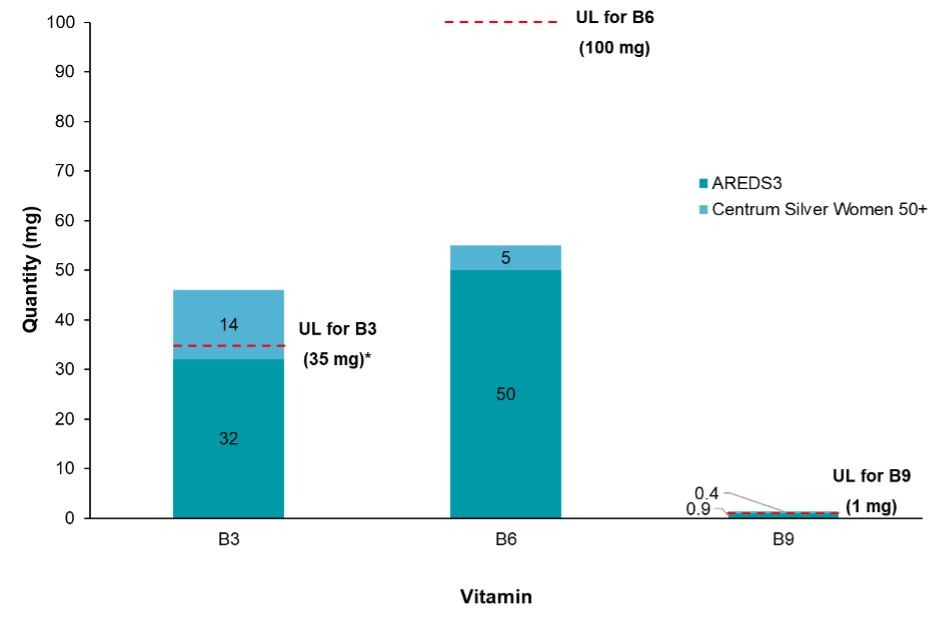
*The UL of 35 mg is based on niacin. Even though the combined intake of vitamin B3 (niacinamide) in AREDS3 and a multivitamin exceeds 35 mg, it is well within the known safety limit for niacinamide (<1500 mg).30
UL, tolerable upper intake level.
The AREDS3 dose of zinc was reduced from 80 mg (AREDS2) to 25 mg, reflecting evidence of similar efficacy at the lower dose.32 When combined with a standard multivitamin, cumulative zinc intake may approach the 40-mg UL but would nonetheless fall well below the 80-mg dose included in AREDS2.
Practical Guidance for Coadministration
Step 1: Assess
Before initiating PreserVision AREDS3, briefly assess the patient’s current supplement and dietary habits. Are they taking a multivitamin, an additional B-complex, receiving B12 injections, or consuming B vitamin–fortified energy drinks regularly?
Step 2: Compare with UL
Next, compare their total estimated B-vitamin intake against the National Institutes of Health Office of Dietary Supplements (NIH/ODS) ULs. Most patients will remain comfortably below these thresholds.
Step 3: Identify exceptions
Identify exceptions who warrant closer review, including those individuals using high-dose standalone B-complex supplements, patients with renal impairment (who may accumulate B6 or have reduced clearance), and individuals who self-escalate supplements without guidance.
Step 4: Counsel
Finally, counsel accordingly. Reassure the majority of users that AREDS3 is safe alongside a standard diet or basic multivitamin, but adjust the regimen for patients clearly exceeding the UL by recommending that they discontinue redundant B-complex products before starting AREDS3.
This structured approach supports safe, individualized use without unnecessarily discouraging appropriate supplementation.
Conclusion
AREDS3 builds on the well-established AREDS2 foundation while adding a B vitamin complex with fully transparent labeling. Notably, the 50 mg of vitamin B6 represents 50% of the NIH/ODS UL, and when total cumulative intake from diet and concurrent supplements is considered, most patients remain within established UL thresholds and well below levels associated with safety concerns. Only a small subset of patients (eg, high-dose B-complex users or those with special medical conditions) would require adjustment. Coadministration with a standard multivitamin or typical diet is manageable through a simple, structured clinical assessment. By grounding counseling in transparent formulation data and established UL benchmarks, ECPs are well-positioned to guide patients toward this advanced formulation with both clinical confidence and evidence-based reassurance.
Julie Poteet, OD, is from MyEyeDr in Acworth, Georgia. She can be reached at drjuliepoteet@gmail.com. Disclosures: Dr. Poteet reports serving as an occasional consultant and advisory board committee member for Bausch + Lomb.
Jessilin Quint, OD, is from Smart Eye Care in Maine. She can be reached at quint.jessilin@gmail.com. Disclosures: Dr. Quint reports serving as an occasional consultant and advisory board committee member for Bausch + Lomb.
Shane R. Kannarr, OD, is from Kannarr Eye Care in Kansas. He can be reached at skannarr@kannarreyecare.com. Disclosures: Dr. Kannarr reports serving as an occasional consultant and advisory board committee member for Bausch + Lomb.
References
- Poteet J, Koetting C, Vakharia PS. Role of B vitamins in preventing the development and progression of age-related macular degeneration. Ophthalmol Ther. 2026;15(1):1-19.
- Elsherbiny NM, Sharma I, Kira D, et al. Homocysteine induces inflammation in retina and brain. Biomolecules. 2020;10(3):393.
- Luo J, Chen X, Yang Y, Liu Y, Feng Y, Chen G. Association of MTHFR C667T polymorphism, homocysteine, and B vitamins with senile cataract. J Nutr Sci Vitaminol (Tokyo). 2023;69(2):136-144.
- Ibrahim AS, Mander S, Hussein KA, et al. Hyperhomocysteinemia disrupts retinal pigment epithelial structure and function with features of age-related macular degeneration. Oncotarget. 2016;7(8):8532-8545.
- Mohamed R, Sharma I, Ibrahim AS, et al. Hyperhomocysteinemia alters retinal endothelial cells barrier function and angiogenic potential via activation of oxidative stress. Sci Rep. 2017;7(1):11952.
- Gu J, Lei C, Zhang M. Folate and retinal vascular diseases. BMC Ophthalmol. 2023;23(1):413.
- Mishra S, Stierman B, Gahche JJ, Potischman N. Dietary supplement use among adults: United States, 2017-2018. NCHS Data Brief. 2021;399:1-8.
- Tan ECK, Eshetie TC, Gray SL, Marcum ZA. Dietary supplement use in middle-aged and older adults. J Nutr Health Aging. 2022;26(2):133-138.
- Scott JM. Folate and vitamin B12. Proc Nutr Soc. 1999;58(2):441-448.
- Hanna M, Jaqua E, Nguyen V, Clay J. B vitamins: functions and uses in medicine. Perm J. 2022;26(2):89-97.
- Homocysteine Lowering Trialists’ Collaboration. Dose-dependent effects of folic acid on blood concentrations of homocysteine: a meta-analysis of the randomized trials. Am J Clin Nutr. 2005;82(4):806-812.
- Seddon JM, Gensler G, Klein ML, Milton RC. C-reactive protein and homocysteine are associated with dietary and behavioral risk factors for age-related macular degeneration. Nutrition. 2006;22(4):441-443.
- Wesselink E, Koekkoek WAC, Grefte S, Witkamp RF, van Zanten ARH. Feeding mitochondria: potential role of nutritional components to improve critical illness convalescence. Clin Nutr. 2019;38(3):982-995.
- Hazim RA, Paniagua AE, Tang L, et al. Vitamin B3, nicotinamide, enhances mitochondrial metabolism to promote differentiation of the retinal pigment epithelium. J Biol Chem. 2022;298(9):102286.
- Rondanelli M, Gasparri C, Riva A, et al. Diet and ideal food pyramid to prevent or support the treatment of diabetic retinopathy, age-related macular degeneration, and cataracts. Front Med (Lausanne). 2023;10:1168560.
- Baltrusch S. The role of neurotropic B vitamins in nerve regeneration. Biomed Res Int. 2021;2021:9968228.
- Bucolo C, Maugeri G, Giunta S, D’Agata V, Drago F, Romano GL. Corneal wound healing and nerve regeneration by novel ophthalmic formulations based on cross-linked sodium hyaluronate, taurine, vitamin B6, and vitamin B12. Front Pharmacol. 2023;14:1109291.
- Boles NC, Fernandes M, Swigut T, et al. Epigenomic and transcriptomic changes during human RPE EMT in a stem cell model of epiretinal membrane pathogenesis and prevention by nicotinamide. Stem Cell Reports. 2020;14(4):631-647.
- Saini JS, Corneo B, Miller JD, et al. Nicotinamide ameliorates disease phenotypes in a human iPSC model of age-related macular degeneration. Cell Stem Cell. 2017;20(5):635-647.
- Ploessl K, Eifler A, Ramadani A, Schaetzler F, Weber BHF. Nicotinamide and its role in reducing complement activation and oxidative stress in age-related macular degeneration [abstract]. Invest Ophthalmol Vis Sci. 2022;63(7):469.
- Wallace TC, Frankenfeld CL, Frei B, et al. Multivitamin/multimineral supplement use is associated with increased micronutrient intakes and biomarkers and decreased prevalence of inadequacies and deficiencies in middle-aged and older adults in the United States. J Nutr Gerontol Geriatr. 2019;38(4):307-328.
- Bailey RL, Jun S, Murphy L, et al. High folic acid or folate combined with low vitamin B-12 status: potential but inconsistent association with cognitive function in a nationally representative cross-sectional sample of US older adults participating in the NHANES. Am J Clin Nutr. 2020;112(6):1547-1557.
- Jun S, Cowan AE, Bhadra A, et al. Older adults with obesity have higher risks of some micronutrient inadequacies and lower overall dietary quality compared to peers with a healthy weight, National Health and Nutrition Examination Surveys (NHANES), 2011-2014. Public Health Nutr. 2020;23(13):2268-2279.
- Weschenfelder C, Cheung SN, Lieberman HR, Berryman CE, Hennigar SR. Variation in micronutrient intake and adequacy by race, ethnicity, and poverty-to-income ratio in United States children and adults: a descriptive analysis of the National Health and Nutrition Examination Survey. J Nutr. 2025;155(12):4455-4464.
- Naurath HJ, Joosten E, Riezler R, Stabler SP, Allen RH, Lindenbaum J. Effects of vitamin B12, folate, and vitamin B6 supplements in elderly people with normal serum vitamin concentrations. Lancet. 1995;346(8967):85-89.
- Agrón E, Mares J, Clemons TE, et al. Dietary nutrient intake and progression to late age-related macular degeneration in the Age-Related Eye Disease Studies 1 and 2. Ophthalmology. 2021;128(3):425-442.
- Christen WG, Glynn RJ, Chew EY, Albert CM, Manson JE. Folic acid, pyridoxine, and cyanocobalamin combination treatment and age-related macular degeneration in women: the Women’s Antioxidant and Folic Acid Cardiovascular Study. Arch Intern Med. 2009;169(4):335-341.
- Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: The National Academies Press; 1998.
- Andrews KW, Roseland JM, Gusev PA, et al. Analytical ingredient content and variability of adult multivitamin/mineral products: national estimates for the Dietary Supplement Ingredient Database. Am J Clin Nutr. 2017;105(2):526-539.
- Young SL, Gazzard G. The adverse effects of oral niacin/nicotinamide – an overview of reviews. Eye (Lond). 2025;39(16):2852-2859.
- Sun Y, Yu X, Teng Y, Sun Y. Toxic effects of excess vitamins A, B6, and folic acid on the nervous system. Behav Neurol. 2025;2025:7888243.
- Age-Related Eye Disease Study 2 Research G. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: the Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA. 2013;309(19):2005-2015.
- Chew EY, Clemons TE, Agrón E, et al. Long-term outcomes of adding lutein/zeaxanthin and ω-3 fatty acids to the AREDS supplements on age-related macular degeneration progression: AREDS2 report 28. JAMA Ophthalmol. 2022;140(7):692-698.

Contact Info
Grandin Library Building
Six Leigh Street
Clinton, New Jersey 08809


